CASE NAME: Som-Dotson v. Alcon Laboratories, Inc.
CASE NO.: Not specified
JURISDICTION: Superior Court of the State of California, County of Los Angeles
FILED ON: April 1, 2026
CLASS DEFINITION: All persons in the United States who purchased Alcon’s Systane eye drop products labeled “preservative free” within four years prior to the filing of the complaint.
SUMMARY:
According to the complaint, Alcon Laboratories, Inc. is accused of falsely marketing its Systane eye drop products as “preservative free” despite containing boric acid, which allegedly functions as a preservative. The lawsuit claims that this labeling misled consumers into believing the products lacked preservatives, a feature many consumers seek for safety or sensitivity reasons. As a result, the plaintiff alleges that consumers paid a premium for products that did not deliver the advertised benefit.
ALLEGATIONS:
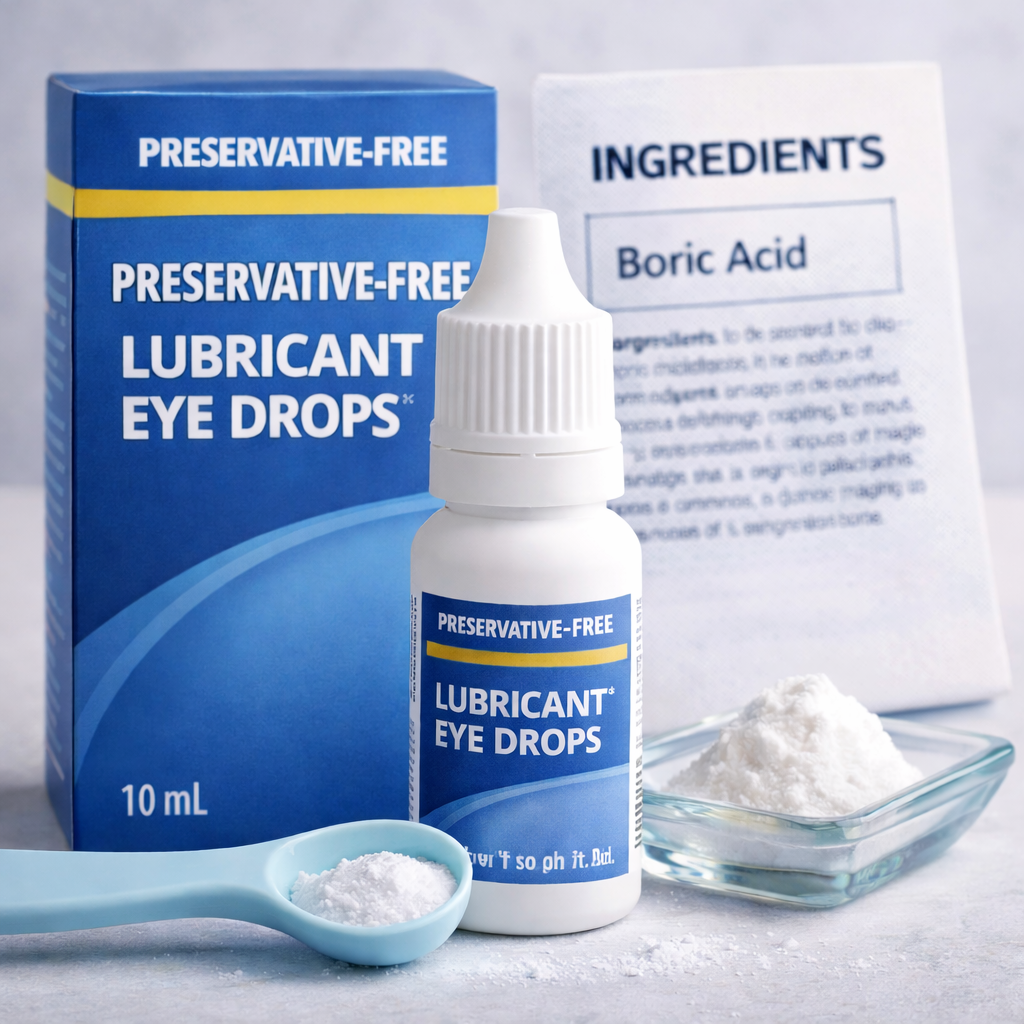
The lawsuit alleges that Alcon manufactures, markets, and sells Systane-branded eye drops nationwide, prominently labeling them as “preservative free.” According to the complaint, this claim is false and misleading because the products contain boric acid, an ingredient alleged to act as a preservative due to its ability to inhibit bacterial growth.
The complaint explains that boric acid has bacteriostatic properties, meaning it can prevent the growth of microorganisms by interfering with their metabolic processes. Because of these properties, the lawsuit alleges that boric acid qualifies as a preservative, contradicting the product’s labeling.
The plaintiff claims she purchased one of the Systane products in September 2025 after reviewing the label, which clearly stated “preservative free” (as depicted on product packaging shown on page 3 of the complaint). She alleges that this representation was a key factor in her purchasing decision, as consumers often seek preservative-free eye drops to avoid irritation or sensitivity.
According to the complaint, reasonable consumers rely on labeling claims to make informed purchasing decisions and are willing to pay more for products that advertise specific health-related benefits, such as being free from preservatives. The lawsuit alleges that Alcon leveraged this preference to increase demand and charge higher prices for its products.
The complaint further alleges that consumers could not reasonably determine that the products contained a preservative ingredient because this information was not clearly disclosed. As a result, consumers were allegedly deprived of the benefit of their bargain—purchasing products they believed were preservative free when they were not.
In addition to economic harm, the plaintiff claims she suffered frustration, loss of trust in product labeling, and wasted time. The lawsuit also alleges that Alcon’s conduct harmed competition by drawing consumers away from other products that were accurately labeled.
The complaint brings claims under California’s False Advertising Law and Unfair Competition Law, alleging that Alcon engaged in unlawful, unfair, and fraudulent business practices. Specifically, the lawsuit asserts that the company knowingly misrepresented the nature of its products and failed to disclose material information necessary for consumers to make informed decisions.
The plaintiff seeks damages, restitution, injunctive relief requiring corrective advertising, and an order preventing Alcon from continuing the alleged misleading practices.






Leave a Reply